Radiation absorption coefficient by mass of material
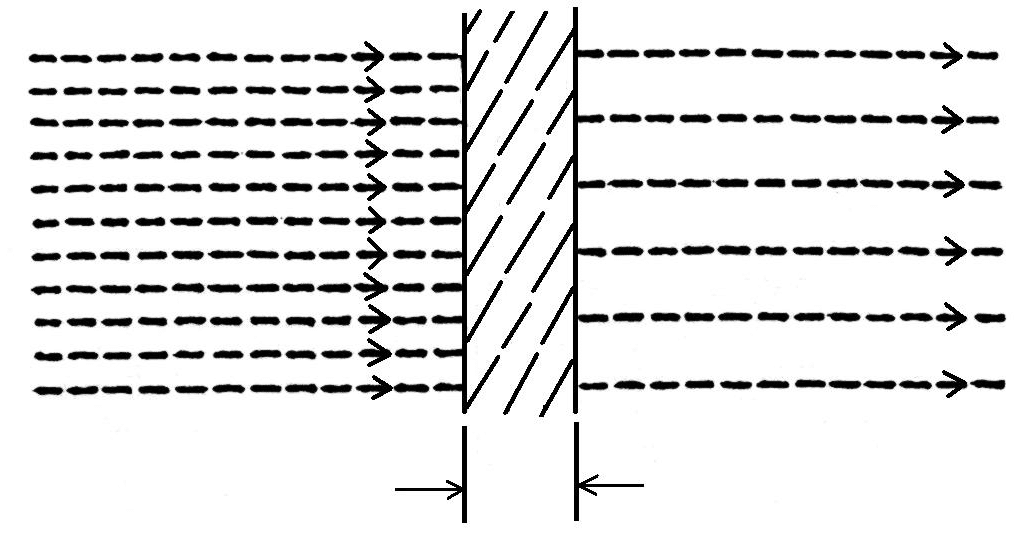
When a beam of X-ray or gamma-ray radiation passes through matter, some rays are transmitted, some are absorbed, and some are scattered in different directions. An understanding of these phenomena is important for a radiographer and its various aspects are outlined below:
Absorption phenomenon
When a beam of X-ray or gamma-ray radiation passes through matter, its intensity is reduced. This phenomenon is called the absorption of X-ray or gamma-ray radiation in matter. The amount of radiation attenuated depends on the quality of the beam, the material, the density of the specimen, and the thickness of the specimen through which the beam passes. This property of X-ray or gamma-ray radiation is used in industrial radiography. If there is a defect within the structure of a specimen, it means that there is a change in thickness (such as voids) or a change in density (such as impurities of other materials in the sample). outside). The presence of these defects induces corresponding changes in the intensity of the transmitted radiation beam and this transmitted radiation beam is recorded on the film producing a radiograph on the film. This phenomenon has a very important property that we need to consider in detail.
The absorbance
In the absorption equation, m is called the linear absorption coefficient. The linear absorption coefficient is the fraction of the radiation intensity that is attenuated per unit thickness of the absorber. The thickness of the absorber is usually given in centimeters (cm) and m .x has no units so m will have units of (cm-1). The distance of 1/m is sometimes called the mean free path of radiation, and to calculate penetration depth is usually expressed in terms of the recovery length and where x = 1/m ; mx = 1 is called a rebound length. Value of m = Kl3Z3 shows that m depends on the wavelength of the primary radiation and therefore soft or low-energy radiation will have a larger absorption coefficient. m also depends on the atomic number (Z) of the absorber and increases with the atomic number (Z). Therefore materials with high atomic number (Z) will absorb more X-ray or gamma-ray radiation than those with low atomic number (Z). In the equation for m, K is a constant that depends on the physical density of the absorber.
Thickness weakens half
Half value layer (HVL) is the thickness of a given material that will reduce the intensity of the emitted radiation beam when passing through it. The HVL half absorption thickness is determined from the formula:
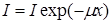
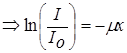
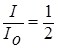

I guess
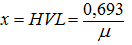
Thickness reduced by 10
10 TVL (Ten value layer) is the thickness of the shielding material that reduces the radiation intensity or radiation dose by 10 times. Calculating similarly to the half absorption thickness we have:

Convert from mass attenuation to linear attenuation
Since the linear attenuation factor (m) depends on the material density, a mass attenuation factor is often used. The mass attenuation factor is simply the linear attenuation factor divided by the material density. This ratio has a constant value for a particular material or compound. To convert the attenuation coefficient by mass (m/r) to the linear attenuation factor (m), simply multiply it by the density (r) of the material.
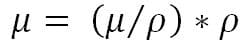
Constants for monatomic materials
Tables and graphs of photon energy attenuation coefficients for the mass of materials from 1 keV to 20 MeV for all molecular masses from Z = 1 to 92, and for compounds and mixtures taken directly from website of the US National Institute of Standards and Technology. The matter constants assume that the material is monatomic. The values are calculated by the atomic number to weight ratio Z/A, the average excitation energy I and the density ρ. Some density values are nominal;
| Atomic Mass (Z) | Material | Z/A | |||
| (eV) | (g/cm3) | ||||
| 1 | H | Hydrogen | 0.99212 | ||
| 2 | He | Helium | 0.49968 | 41.8 | 1.663E-04 |
| 3 | Li | Lithium | 0.43221 | 40.0 | 5.340E-01 |
| 4 | Beige | Beryllium | 0.44384 | 63.7 | 1.848E+00 |
| 5 | REMOVE | Boron | 0.46245 | 76.0 | 2.370E+00 |
| 6 | OLD | Carbon, Graphite | 0.49954 | 78.0 | 1.700E+00 |
| 7 | WOMEN | Nitrogen | 0.49976 | 82.0 | 1.165E-03 |
| 8 | O | Oxygen | 0.50002 | 95.0 | 1.332E-03 |
| 9 | F | Fluorine | 0.47372 | 115.0 | 1.580E-03 |
| 10 | Ne | neon | 0.49555 | 137.0 | 8.385E-04 |
| 11 | Na | Sodium | 0.47847 | 149.0 | 9.710E-01 |
| 12 | Mg | Magnesium | 0.49373 | 156.0 | 1.740E+00 |
| 13 | Al | Aluminum | 0.48181 | 166.0 | 2.699E+00 |
| 14 | Si | Silicon | 0.49848 | 173.0 | 2.330E+00 |
| 15 | P | Phosphorus | 0.48428 | 173.0 | 2.200E+00 |
| 16 | S | Sulfur | 0.49897 | 180.0 | 2.000E+00 |
| 17 | Cl | Chlorine | 0.47951 | 174.0 | 2.995E-03 |
| 18 | Ar | Argon | 0.45059 | 188.0 | 1.662E-03 |
| 19 | KY | Potassium | 0.48595 | 190.0 | 8.620E-01 |
| 20 | Shift | Calcium | 0.49903 | 191.0 | 1.550E+00 |
| 21 | Sc | Scandium | 0.46712 | 216.0 | 2.989E+00 |
| 22 | Ti | Titanium | 0.45948 | 233.0 | 4.540E+00 |
| 23 | DRAW | Vanadium | 0.45150 | 245.0 | 6.110E+00 |
| 24 | Cr | Chromium | 0.46157 | 257.0 | 7.180E+00 |
| 25 | Mn | Manganese | 0.45506 | 272.0 | 7.440E+00 |
| 26 | Fe | Iron | 0.46556 | 286.0 | 7.874E+00 |
| 27 | Co | Cobalt | 0.45815 | 297.0 | 8.900E+00 |
| 28 | Ni | Nickel | 0.47708 | 311.0 | 8.902E+00 |
| 29 | Cu | Copper | 0.45636 | 322.0 | 8.960E+00 |
| 30 | Zn | Zinc | 0.45879 | 330.0 | 7.133E+00 |
| 31 | Ga | Gallium | 0.44462 | 334.0 | 5.904E+00 |
| 32 | Ge | germanium | 0.44071 | 350.0 | 5.323E+00 |
| 33 | As | Arsenal | 0.44046 | 347.0 | 5.730E+00 |
| 34 | Se | Selenium | 0.43060 | 348.0 | 4.500E+00 |
| 35 | Br | Bromine | 0.43803 | 343.0 | 7.072E-03 |
| 36 | Kr | Krypton | 0.42959 | 352.0 | 3.478E-03 |
| 37 | Rb | Rubidium | 0.43291 | 363.0 | 1.532E+00 |
| 38 | Sr | Strontium | 0.43369 | 366.0 | 2.540E+00 |
| 39 | Y | Yttrium | 0.43867 | 379.0 | 4.469E+00 |
| 40 | Zr | Zirconium | 0.43848 | 393.0 | 6.506E+00 |
| 41 | Nb | Niobium | 0.44130 | 417.0 | 8.570E+00 |
| 42 | Mo | Molybdenum | 0.43777 | 424.0 | 1.022E+01 |
| 43 | Tc | Technetium | 0.43919 | 428.0 | 1.150E+01 |
| 44 | Ru | Ruthenium | 0.43534 | 441.0 | 1.241E+01 |
| 45 | Rh | Rhodium | 0.43729 | 449.0 | 1.241E+01 |
| 46 | Pd | Palladium | 0.43225 | 470.0 | 1.202E+01 |
| 47 | Ag | silver | 0.43572 | 470.0 | 1.050E+01 |
| 48 | CD | Cadmium | 0.42700 | 469.0 | 8.650E+00 |
| 49 | Indium | 0.42676 | 488.0 | 7.310E+00 | |
| 50 | SN | Believe | 0.42120 | 488.0 | 7.310E+00 |
| 51 | Sb | Antimony | 0.41889 | 487.0 | 6.691E+00 |
| 52 | Te | Tellurium | 0.40752 | 485.0 | 6.240E+00 |
| 53 | I | Iodine | 0.41764 | 491.0 | 4.930E+00 |
| 54 | Car | Xenon | 0.41130 | 482.0 | 5.485E-03 |
| 55 | Cs | Cesium | 0.41383 | 488.0 | 1.873E+00 |
| 56 | three | Barium | 0.40779 | 491.0 | 3.500E+00 |
| 57 | La | Lanthanum | 0.41035 | 501.0 | 6.154E+00 |
| 58 | Ce | Cerium | 0.41395 | 523.0 | 6.657E+00 |
| 59 | Pr | Praseodymium | 0.41871 | 535.0 | 6.710E+00 |
| 60 | Nd | Neodymium | 0.41597 | 546.0 | 6.900E+00 |
| 61 | Pm | Promethium | 0.42094 | 560.0 | 7.220E+00 |
| 62 | Sm | Samarium | 0.41234 | 574.0 | 7.460E+00 |
| 63 | Eu | Europium | 0.41457 | 580.0 | 5.243E+00 |
| 64 | Gd | Gadolinium | 0.40699 | 591.0 | 7.900E+00 |
| 65 | Tb | Terbium | 0.40900 | 614.0 | 8.229E+00 |
| 66 | Dy | Dysprosium | 0.40615 | 628.0 | 8.550E+00 |
| 67 | A cough | Holmium | 0.40623 | 650.0 | 8.795E+00 |
| 68 | Er | Erbium | 0.40655 | 658.0 | 9.066E+00 |
| 69 | Tm | Thulium | 0.40844 | 674.0 | 9.321E+00 |
| 70 | YB | Ytterbium | 0.40453 | 684.0 | 6.730E+00 |
| 71 | Lu | Lutetium | 0.40579 | 694.0 | 9.840E+00 |
| 72 | Hf | Hafnium | 0.40338 | 705.0 | 1.331E+01 |
| 73 | I | Tantalum | 0.40343 | 718.0 | 1.665E+01 |
| 74 | W | Tungsten | 0.40250 | 727.0 | 1.930E+01 |
| 75 | Re | Rhenium | 0.40278 | 736.0 | 2.102E+01 |
| 76 | Os | Osmium | 0.39958 | 746.0 | 2.257E+01 |
| 77 | Ir | Iridium | 0.40058 | 757.0 | 2.242E+01 |
| 78 | PT | Platinum | 0.39984 | 790.0 | 2.145E+01 |
| 79 | Au | Gold | 0.40108 | 790.0 | 1.932E+01 |
| 80 | Hg | Mercury | 0.39882 | 800.0 | 1.355E+01 |
| 81 | Tl | Thallium | 0.39631 | 810.0 | 1.172E+01 |
| 82 | Pb | Lead | 0.39575 | 823.0 | 1.135E+01 |
| 83 | Bi | Bismuth | 0.39717 | 823.0 | 9.747E+00 |
| 84 | Po | Polonium | 0.40195 | 830.0 | 9.320E+00 |
| 85 | At | Astatine | 0.40479 | 825.0 | 1.000E+01 |
| 86 | Rn | Radon | 0.38736 | 794.0 | 9.066E-03 |
| 87 | Fr | francium | 0.39010 | 827.0 | 1.000E+01 |
| 88 | Out | Radium | 0.38934 | 826.0 | 5.000E+00 |
| 89 | Ac | Actinium | 0.39202 | 841.0 | 1.007E+01 |
| 90 | Th | Thorium | 0.38787 | 847.0 | 1.172E+01 |
| 91 | Pa | Protactinium | 0.39388 | 878.0 | 1.537E+01 |
| 92 | U | Uranium | 0.38651 | 890.0 | 1.895E+01 |
Absorption (attenuation) constant for composite materials
The material constant with composition is assumed for compounds and complex mixtures. The various human tissue components were obtained from ICRU Report 44 (1989). The values are calculated by the atomic number to weight ratio Z/A, the average excitation energy I and the density ρ. Some density values are nominal;
| Material | <Z/A> | I | ||
| Adipose Tissue (ICRU-44) | 0.55579 | 64.8 | 9.500E-01 | 1: 0.114000 6: 0.598000 7: 0.007000 8: 0.278000 11: 0.001000 16: 0.001000 17: 0.001000 |
| Air, Dry (near sea level) | 0.49919 | 85.7 | 1.205E-03 | 6: 0.000124 7: 0.755268 8: 0.231781 18: 0.012827 |
| Alanine | 0.53876 | 71.9 | 1.424E+00 | 1: 0.079192 6: 0.404437 7: 0.157213 8: 0.359157 |
| B-100 Bone-Equivalent Plastic | 0.52740 | 85.9 | 1.450E+00 | 1: 0.065473 6: 0.536942 7: 0.021500 8: 0.032084 9: 0.167415 20: 0.176585 |
| Bakelite | 0.52792 | 72.4 | 1.250E+00 | 1: 0.057444 6: 0.774589 8: 0.167968 |
| Blood, Whole (ICRU-44) | 0.54999 | 75.2 | 1.060E+00 | 1: 0.102000 6: 0.110000 7: 0.033000 8: 0.745000 11: 0.001000 15: 0.001000 16: 0.002000 17: 0.003000 19: 0.002000 26: 0.001000 |
| Bone, Cortical (ICRU-44) | 0.51478 | 112.0 | 1.920E+00 | 1: 0.034000 6: 0.155000 7: 0.042000 8: 0.435000 11: 0.001000 12: 0.002000 15: 0.103000 16: 0.003000 20: 0.225000 |
| Brain, Gray/White Matter (ICRU-44) | 0.55239 | 73.9 | 1.040E+00 | 1: 0.107000 6: 0.145000 7: 0.022000 8: 0.712000 11: 0.002000 15: 0.004000 16: 0.002000 17: 0.003000 19: 0.003000 |
| Breast Tissue (ICRU-44) | 0.55196 | 70.3 | 1.020E+00 | 1: 0.106000 6: 0.332000 7: 0.030000 8: 0.527000 11: 0.001000 15: 0.001000 16: 0.002000 17: 0.001000 |
| C-552 Air-equivalent Plastic | 0.49969 | 86.8 | 1.760E+00 | 1: 0.024681 6: 0.501610 8: 0.004527 9: 0.465209 14: 0.003973 |
| Cadmium Telluride | 0.41665 | 539.3 | 6.200E+00 | 48: 0.468358 52: 0.531642 |
| Calcium Fluoride | 0.48671 | 166.0 | 3.180E+00 | 9: 0.486672 20: 0.513328 |
| Calcium Sulfate | 0.49948 | 152.3 | 2.960E+00 | 8: 0.470081 16: 0.235534 20: 0.294385 |
| 15 mmol L-1 Ceric Ammonium Sulfate Solution | 0.55282 | 76.7 | 1.030E+00 | 1: 0.107694 7: 0.000816 8: 0.875172 16: 0.014279 58: 0.002040 |
| Cesium Iodide | 0.41569 | 553.1 | 4.510E+00 | 53: 0.488451 55: 0.511549 |
| Concrete, Ordinary | 0.50932 | 124.5 | 2.300E+00 | 1: 0.022100 6: 0.002484 8: 0.574930 11: 0.015208 12: 0.001266 13: 0.019953 14: 0.304627 19: 0.010045 20: 0.042951 26: 0.006435 |
| Concrete, Barite (TYPE BA) | 0.45714 | 248.2 | 3.350E+00 | 1: 0.003585 8: 0.311622 12: 0.001195 13: 0.004183 14: 0.010457 16: 0.107858 20: 0.050194 26: 0.047505 56: 0.463400 |
| Eye Lens (ICRU-44) | 0.54709 | 74.3 | 1.070E+00 | 1: 0.096000 6: 0.195000 7: 0.057000 8: 0.646000 11: 0.001000 15: 0.001000 16: 0.003000 17: 0.001000 |
| Ferrous Sulfate Standard Fricke | 0.55334 | 76.3 | 1.024E+00 | 1: 0.108376 8: 0.878959 11: 0.000022 16: 0.012553 17: 0.000035 26: 0.000055 |
| Gadolinium Oxysulfide | 0.42265 | 493.3 | 7.440E+00 | 8: 0.084527 16: 0.084704 64: 0.830769 |
| Gafchromic Sensor | 0.54384 | 67.2 | 1.300E+00 | 1: 0.089700 6: 0.605800 7: 0.112200 8: 0.192300 |
| Gallium Arsenide | 0.44246 | 384.9 | 5.310E+00 | 31: 0.482030 33: 0.517970 |
| Glass, Borosilicate (Pyrex) | 0.49707 | 134.0 | 2.230E+00 | 5: 0.040066 8: 0.539559 11: 0.028191 13: 0.011644 14: 0.377220 19: 0.003321 |
| Glass, Lead | 0.42101 | 526.4 | 6.220E+00 | 8: 0.156453 14: 0.080866 22: 0.008092 33: 0.002651 82: 0.751938 |
| Lithium Fluoride | 0.46262 | 94.0 | 2.635E+00 | 3: 0.267585 9: 0.732415 |
| Lithium Tetraborate | 0.48485 | 94.6 | 2.440E+00 | 3: 0.082081 5: 0.255715 8: 0.662204 |
| Lung Tissue (ICRU-44) | 0.55048 | 75.2 | 1.050E+00 | 1: 0.103000 6: 0.105000 7: 0.031000 8: 0.749000 11: 0.002000 15: 0.002000 16: 0.003000 17: 0.003000 19: 0.002000 |
| Magnesium Tetroborate | 0.49012 | 108.3 | 2.530E+00 | 5: 0.240870 8: 0.623762 12: 0.135367 |
| Mercuric Iodide | 0.40933 | 684.5 | 6.360E+00 | 53: 0.558560 80: 0.441440 |
| Muscle, Skeletal (ICRU-44) | 0.55000 | 74.6 | 1.050E+00 | 1: 0.102000 6: 0.143000 7: 0.034000 8: 0.710000 11: 0.001000 15: 0.002000 16: 0.003000 17: 0.001000 19: 0.004000 |
| Ovary (ICRU-44) | 0.55149 | 75.0 | 1.050E+00 | 1: 0.105000 6: 0.093000 7: 0.024000 8: 0.768000 11: 0.002000 15: 0.002000 16: 0.002000 17: 0.002000 19: 0.002000 |
| Photographic Emulsion (Kodak Type AA) | 0.48176 | 179.0 | 2.200E+00 | 1: 0.030500 6: 0.210700 7: 0.072100 8: 0.163200 35: 0.222800 47: 0.300700 |
| Photographic Emulsion (Standard Nuclear) | 0.45453 | 331.0 | 3.815E+00 | 1: 0.014100 6: 0.072261 7: 0.019320 8: 0.066101 16: 0.001890 35: 0.349104 47: 0.474105 53: 0.003120 |
| Plastic Scintillator, Vinyltoluene | 0.54141 | 64.7 | 1.032E+00 | 1: 0.085000 6: 0.915000 |
| Polyethylene | 0.57033 | 57.4 | 9.300E-01 | 1: 0.143716 6: 0.856284 |
| Polyethylene Terephthalate, (Mylar) | 0.52037 | 78.7 | 1.380E+00 | 1: 0.041960 6: 0.625016 8: 0.333024 |
| Polymethyl Methacrylate | 0.53937 | 74.0 | 1.190E+00 | 1: 0.080541 6: 0.599846 8: 0.319613 |
| Polystyrene | 0.53768 | 68.7 | 1.060E+00 | 1: 0.077421 6: 0.922579 |
| Polytetrafluoroethylene, (Teflon) | 0.47993 | 99.1 | 2.250E+00 | 6: 0.240183 9: 0.759818 |
| Polyvinyl Chloride | 0.51201 | 108.2 | 1.406E+00 | 1: 0.048382 6: 0.384361 17: 0.567257 |
| Radiochromic Dye Film, Nylon Base | 0.54987 | 64.5 | 1.080E+00 | 1: 0.101996 6: 0.654396 7: 0.098915 8: 0.144693 |
| Testis (ICRU-44) | 0.55200 | 74.7 | 1.040E+00 | 1: 0.106000 6: 0.099000 7: 0.020000 8: 0.766000 11: 0.002000 15: 0.001000 16: 0.002000 17: 0.002000 19: 0.002000 |
| Tissue, Soft (ICRU-44) | 0.54996 | 74.7 | 1.060E+00 | 1: 0.102000 6: 0.143000 7: 0.034000 8: 0.708000 11: 0.002000 15: 0.003000 16: 0.003000 17: 0.002000 19: 0.003000 |
| Tissue, Soft (ICRU Four-Component) | 0.54975 | 74.9 | 1.000E+00 | 1: 0.101174 6: 0.111000 7: 0.026000 8: 0.761826 |
| Tissue-Equivalent Gas, Methane Based | 0.54992 | 61.2 | 1.064E-03 | 1: 0.101873 6: 0.456177 7: 0.035172 8: 0.406778 |
| Tissue-Equivalent Gas, Propane Based | 0.55027 | 59.5 | 1.826E-03 | 1: 0.102676 6: 0.568937 7: 0.035022 8: 0.293365 |
| Water, Liquid | 0.55508 | 75.0 | 1.000E+00 | 1: 0.111898 8: 0.888102 |



























